TITANIUM NITRIDE (TiN) COATINGS
Engineered for Reliability and Long-Term Implant Performance
Pulse Technologies provides titanium nitride (TiN) coatings designed to support efficient charge transfer, electrochemical stability, and long-term biocompatibility in implantable cardiac and neurostimulation devices.
As implantable devices demand increasing reliability and electrochemical efficiency, surface coatings play a critical role in maintaining device performance and biocompatibility.
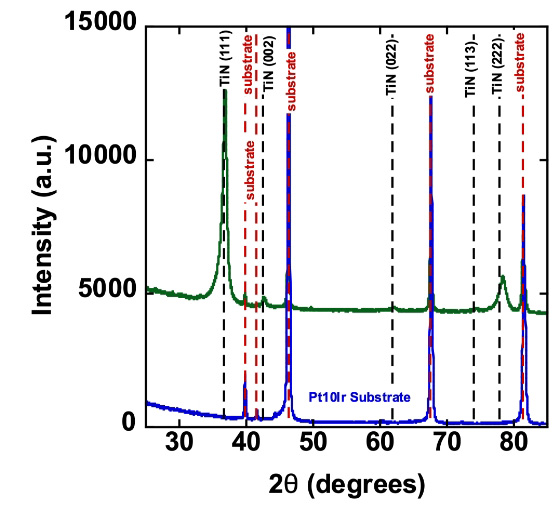
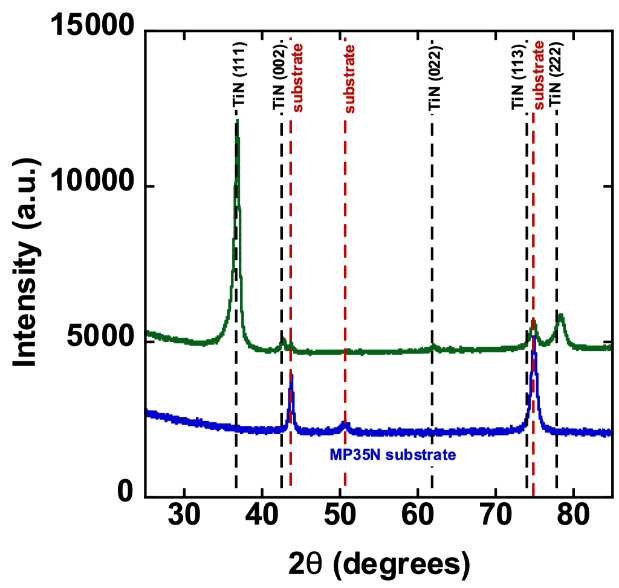
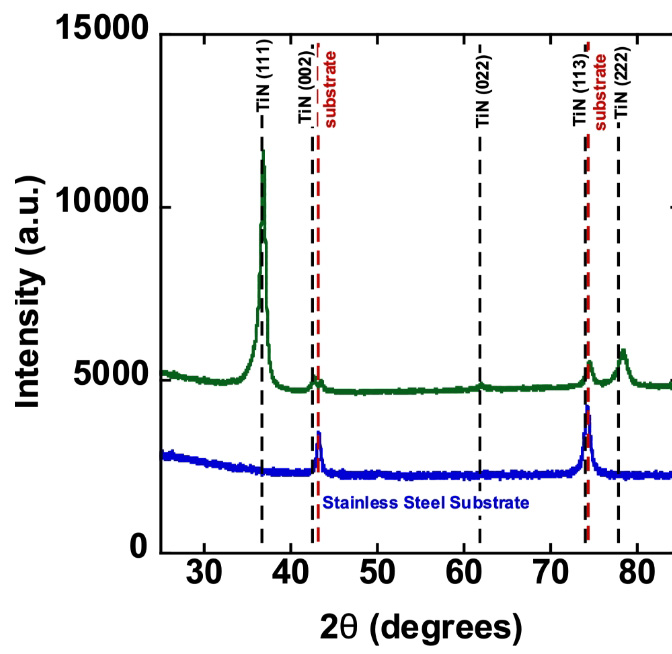
Titanium nitride (TiN) coatings are industry-standard coatings typically deposited by physical vapor deposition (PVD) and are widely recognized for their excellent biocompatibility and favorable electrochemical properties. TiN has a long and well-established track record in implantable medical devices, including cardiac rhythm management systems such as pacemakers, where it provides a stable and durable electrode surface for demanding cardiac and neurostimulation applications.
TiN coatings can be deposited reproducibly across a wide range of electrode geometries. These coatings typically exhibit a columnar, fractal, semi-porous microstructure that increases effective surface area and supports enhanced electrochemical performance.
As a result, TiN-coated electrodes are widely used in cardiac rhythm management (CRM) devices and are also applicable to other neural interfacing applications where stable and efficient charge transfer is required.
Optimizing Energy Efficiency in Cardiac Rhythm Management
The performance of cardiac rhythm management (CRM) devices depends on the energy required to stimulate cardiac tissue. This energy is determined by the programmed pulse width and amplitude, as well as the voltage delivered between the electrodes—the anode and the cathode. Because the electrode surface directly influences how efficiently electrical energy is transferred to cardiac tissue, optimizing the electrode–tissue interface can reduce current drain and improve overall device efficiency. Clinically, improvements in this interface can contribute to extended battery life and longer device longevity.
Electrodes used in CRM devices, such as pacemakers, are metallic conductors that mediate the transition from electronic current within the electrode to ionic current in the surrounding tissue through electrochemical processes occurring at the electrode–tissue interface.
Why Material Choice Defines Device Longevity and Reliability
The materials used in implantable electrodes do more than conduct electrical current—they play a critical role in determining how reliably a device performs during long-term operation within the body.
Selecting materials and surface technologies for implantable electrodes requires careful consideration of material properties, surface microstructure, electrochemical behavior, and charge-transfer characteristics. The electrode surface must efficiently deliver electrical stimulation while also enabling accurate sensing of the cardiac response.
In cardiac rhythm management (CRM) devices, charge transfer ideally occurs through double-layer charging and discharging at the electrode–electrolyte interface. This capacitive charge-exchange mechanism is desirable because it avoids the generation or consumption of chemical species during a stimulation pulse. Achieving high charge-injection capacity therefore depends on electrode surfaces that provide large effective surface area, typically through porous microstructures or high-surface-area coatings.

Titanium nitride (TiN) coatings have long been used on platinum–iridium (Pt/Ir) electrodes in cardiac rhythm management (CRM) applications. TiN is a chemically stable, electrically conductive material with excellent biocompatibility that supports charge injection through the electrode–electrolyte double layer.
Electrodes incorporating high-surface-area TiN coatings can therefore achieve high charge-injection capacities. The surface microstructure of TiN coatings can also promote controlled tissue in-growth at the electrode–tissue interface, which may contribute to improved long-term stability and device performance.
At Pulse Technologies, TiN coatings are applied using precision physical vapor deposition (PVD) processes supported by extensive experience in implantable device manufacturing. These capabilities enable consistent coating quality, controlled surface morphology, and reliable electrochemical performance for OEM partners developing cardiac rhythm management and neurostimulation devices.

Engineered for Reliability and Long-Term Implant Performance
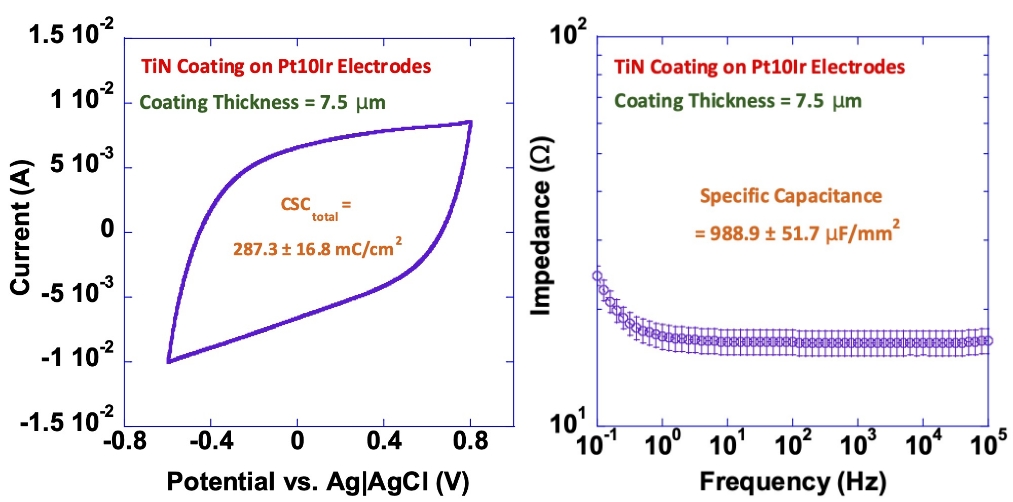
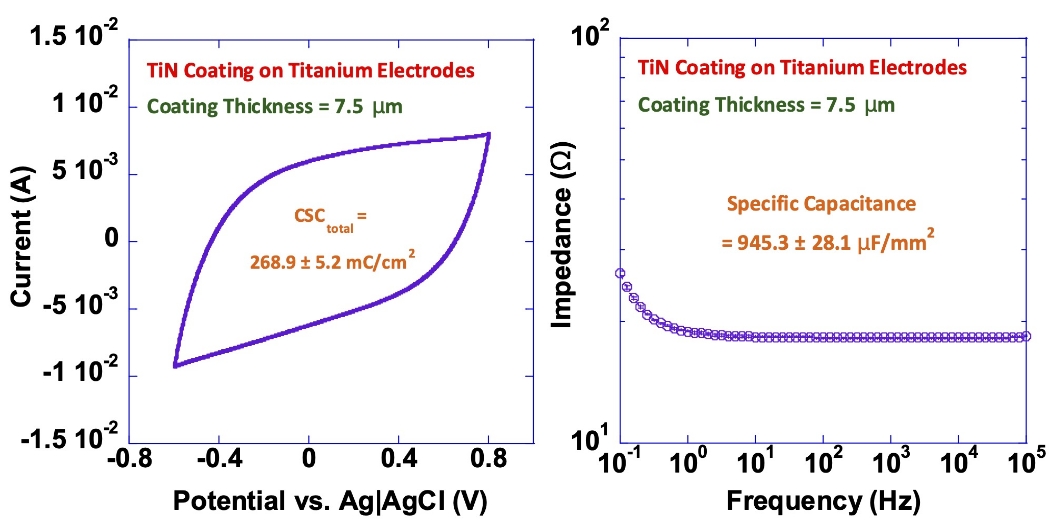
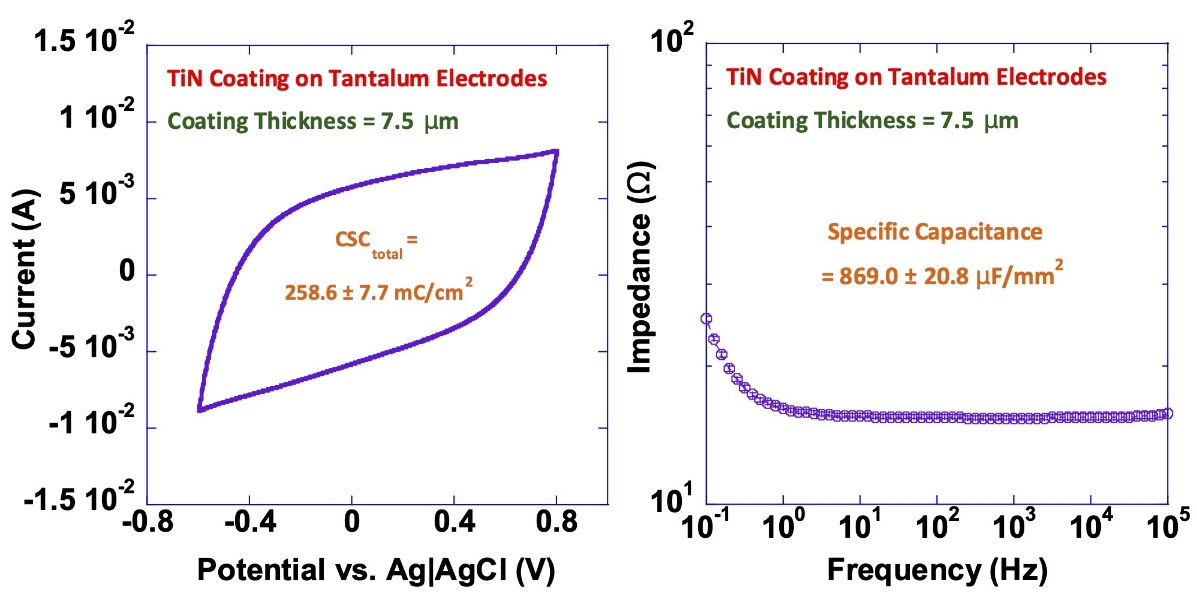
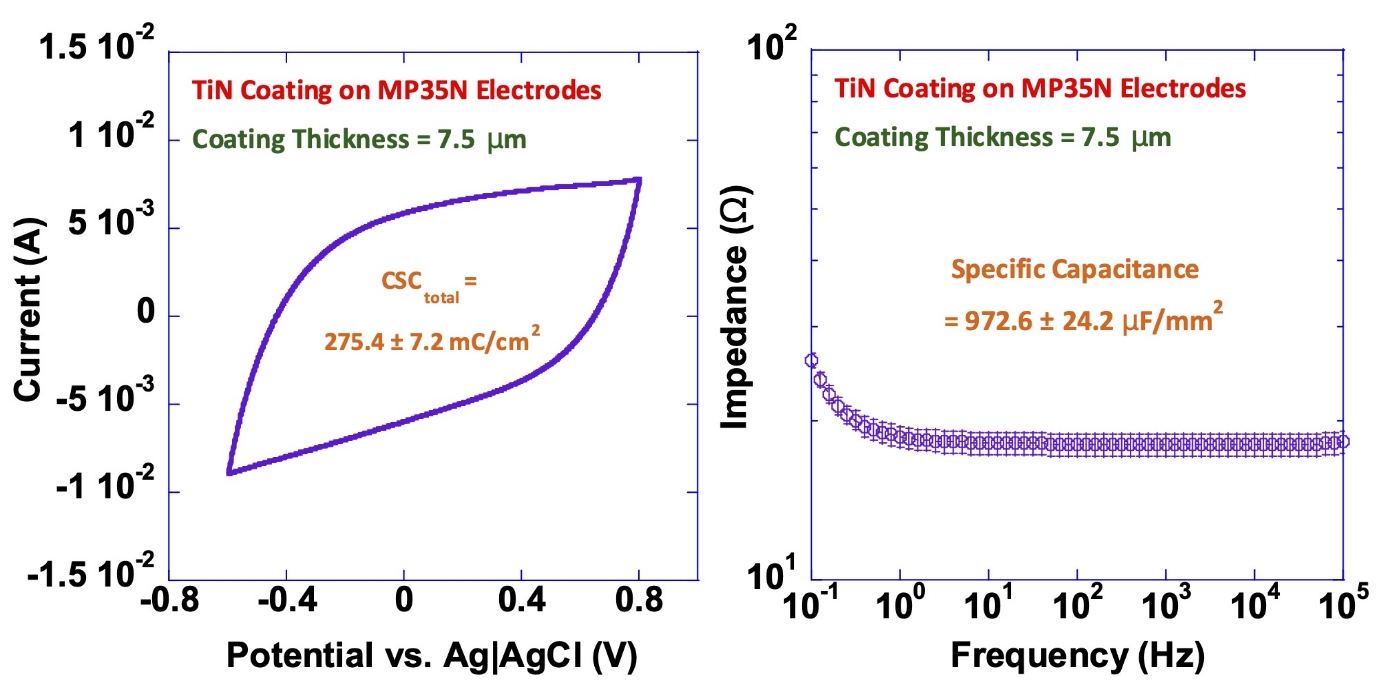
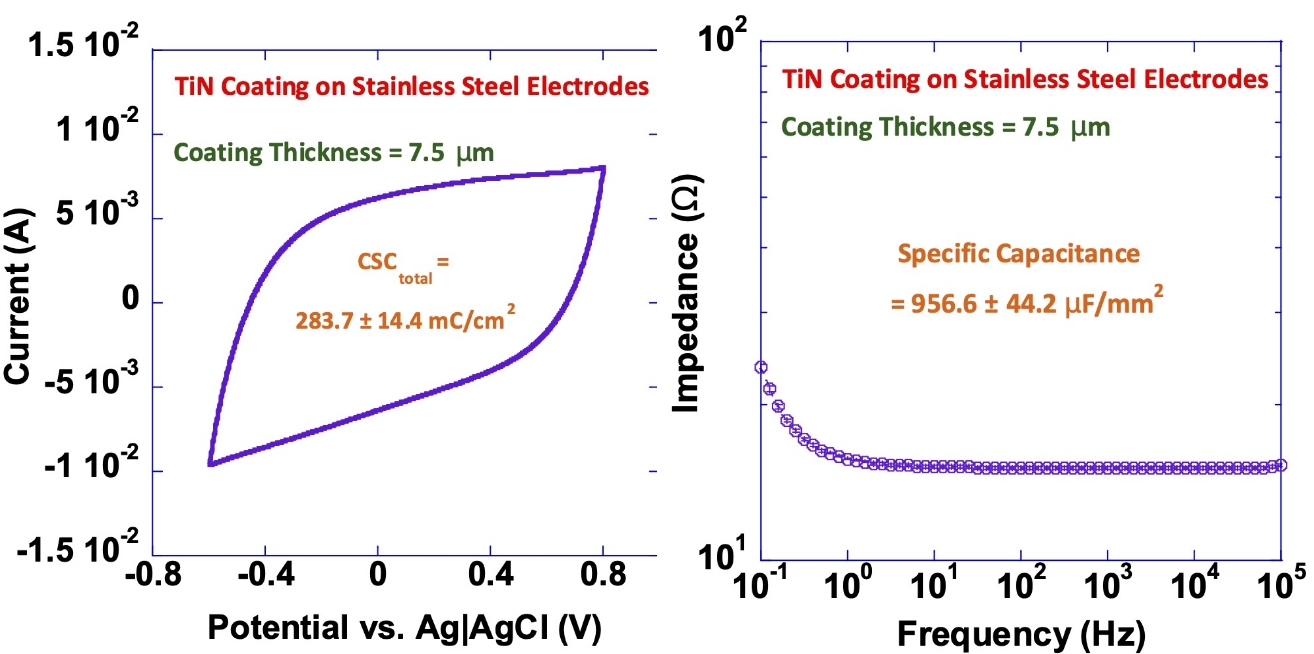
Our titanium nitride (TiN) coatings provide high capacitance and charge-injection capacity while reducing electrode impedance.
The diagrams below illustrate these effects, showing the increased charge-storage capacity (left) and lower impedance (right) achieved with TiN-coated electrodes.
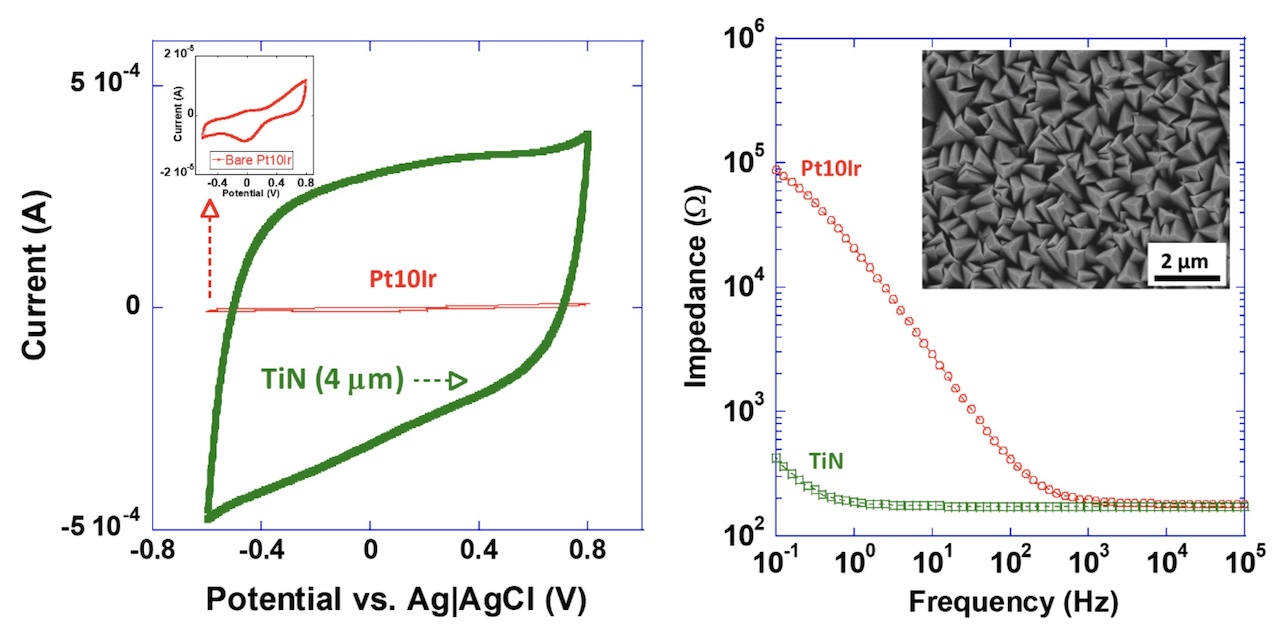
These results demonstrate the measurable performance advantages of TiN coatings in long-term implantable applications.
TiN Coating on Pt10Ir Electrodes


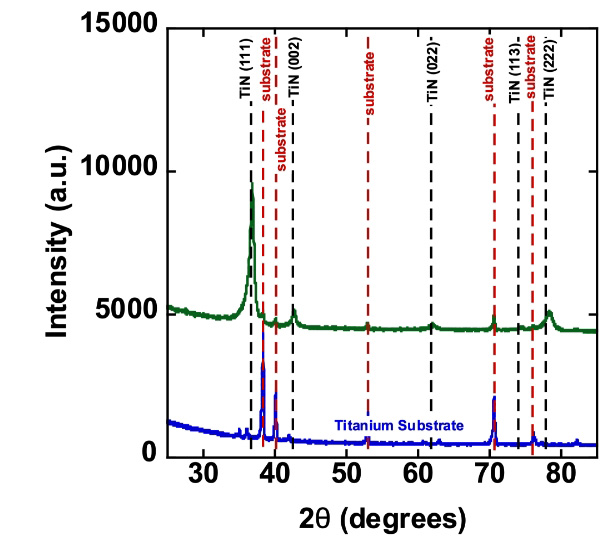
TiN Coating on Titanium Electrodes


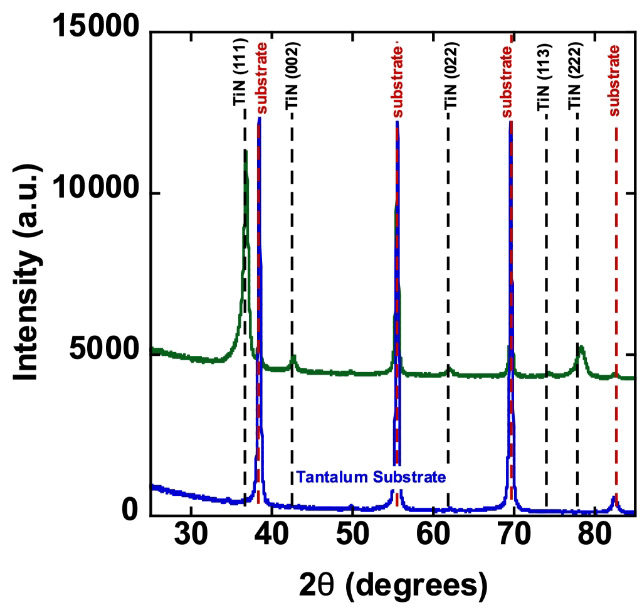
TiN Coating on Tantalum Electrodes


TiN Coating on MP35N Electrodes


TiN Coating on Stainless Steel Electrodes


Ready to explore how Pulse Technologies can help you achieve these performance gains in your next device program?
Recent News & Blogs on HSR™
Femtosecond laser hierarchical surface restructuring for next generation neural interfacing electrodes and microelectrode arrays
Shahram Amini, Wesley Seche, Nicholas May, Hongbin Choi, Pouya Tavousi & Sina Shahbazmohamadi Long-term implantable neural interfacing devices are able to diagnose, monitor, and treat many cardiac, neurological, retinal and hearing disorders through nerve...
Pulse Technologies Announces Successful Biocompatibility Results For Novel HSR™ Platinum-Iridium Electrode Surface Treatment Technology
Biocompatibility studies were conducted by an independent, certified lab based in the US in accordance with FDA and ISO standards.
Surface Treatment Technology Using Ultrashort Pulse Lasers Could Help Treat Heart Failure
Hierarchical Surface Restructuring technology can be used to enhance charge storage capacity, increase specific capacitance, & decrease impedance of electrodes & microelectrodes.
Pulse Technologies’ Electrode Surface Treatment Chosen for Cardiac Device
Pulse Technologies’ HSR™ electrode surface treatment is now part of Cardionomic’s Cardiac Pulmonary Nerve Stimulation (CPNS) System for treating heart failure.
Hierarchically Restructured Titanium Electrodes: A Novel, Low-Cost, High-Performing Platform for Leadless Pacemakers
Pulse’s hierarchical titanium surface restructuring is well-suited for enhancing titanium electrodes of leadless pacemakers.
How Pulse’s Newly Patented Hierarchical Surface Restructuring (HSR™) Technology Improves Electrode Performance in Implantable and Diagnostic Medical Devices
This blog post outlines how Pulse’s patented HSR™ technology can enhance the electrochemical performance of electrodes and microelectrode arrays via enhancements in their surface topology and surface characteristics.
Video Explains the Benefits of Hierarchical Surface Restructuring
This video, Hierarchical Surface Restructuring for Electrodes And Microelectrode Arrays, will introduce you to a unique technology that uses lasers to rearrange the molecular surface of electrode materials and promises to enhance the performance of next-generation sensing, recording and stimulating devices.
Contract Manufacturing
Advanced Technology
Capabilities
© 2026 Pulse Technologies, Inc. All rights reserved.
 Careers
Careers Contact
Contact










